Abstract
INTRODUCTION: Although the treatment of patients with multiple myeloma (MM) has dramatically improved, those with high-risk characteristics, including the deletion or mutation of the master tumor suppressor gene TP53 on chromosome 17, experience limited survival. OM301 is a synthetic polypeptide containing the p53TA (transactivation) domain, which prevents p53 degradation through inhibition of MDM2. Here, we demonstrate that OM301 has strong anti-MM activity in vitro and in vivo.
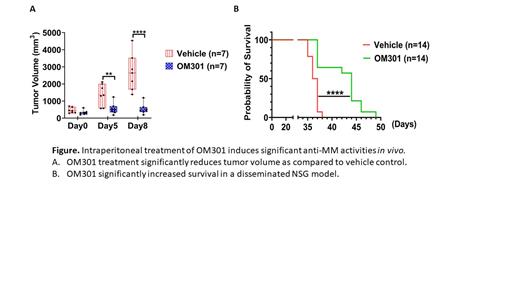
RESULTS: We first assessed the cytotoxic effects of OM301 in MM cell lines with varying TP53 status (TP53 wild type: MM.1S, H929; TP53 mutated/null: L363, RPMI-8226, U266, JJN3, KMS11) and found that OM301 exerts significant cytotoxic effects at a concentration of ~5 µM in all cell lines we tested, while it was minimally toxic to human peripheral blood mononuclear cells. Next, using immunocompromised NSG mice models injected with MM.1S, we determined the in vivo efficacy of OM301 in three different studies. Many potent anticancer agents, particularly of peptide origin, show prominent anti-tumor effects but fail to sustain similar effects when given intraperitoneally because of poor absorption, distribution, metabolism and excretion properties. OM301 at an intraperitoneal dose of 20 mg/kg/body weight twice a day induced significant reduction in tumor size with respect to vehicle control, suggesting the stability of OM301 without any loss of its activity (n=7, p<0.0001). Accordingly, we investigated its effect in a disseminated NSG/MM.1S model and found that it significantly increased survival (p<0.0001) (see Figure).
Because OM301 was designed to simulate the p53 interaction domain with MDM2, we first determined its effect on p53-MDM2 crosstalk using a p53-MDM2 co-Immunoprecipitation (co-IP) assay and compared it with effects from Nutlin-3a, a known inhibitor of p53-MDM2. The co-IP data showed that, unlike Nutlin-3a, OM301 does not inhibit the p53-MDM2 interaction. Thus, to confirm our findings, we first overexpressed MDM2 in HeLa cells, and, using MDM2-IP and p53-MDM2 co-IP, found similar observations. Additionally, OM301 also failed to induce endogenous upregulation of genes activated by p53, such as MDM2 and p21, as opposed to results from Nutlin-3a. RNA sequencing data also showed a distinctive OM301signature, as compared to Nutlin-3a in MM cells. While treatment of Nutlin-3a induced expression of p53-activated canonical genes, OM301-treated cells showed alterations in genes involved in inflammatory responses, c-Myc regulated genes, fatty acid metabolism, glucose metabolism, and oxidative phosphorylation, among others.
Next, to dissect its underlying mechanism, we dual-tagged OM301 with fluorophores at the 3' and 5' ends to study its localization and its stability in MM cells. Indeed, OM301 was found to be stable and mainly localized in the cytosol. We then modified OM301 by biotinylation of its penetratin end and first verified its cytotoxic effect in different MM cell lines, which was similar to that of native OM301. The biotinylated OM301 was then immunoprecipitated using streptavidin beads. The streptavidin pull-down and subsequent proteomic analysis confirmed that OM301 does not interact with MDM2 but interacts with c-Myc and with proteins localized in mitochondria, including Bcl-2 and Bcl-2 family members such as Bclaf1, Bcl2L13, and Bcl2L1. Pull-down experiments and immunoblot analysis validated Bcl-2/OM301 interactions. To further evaluate the relative binding potentials of OM301, we performed molecular docking studies using the HPEPDOCK server (Yan et al., Nat Protoc. 2020;15:1829). Post-docking, the calculated docking scores for OM301 was -281, suggesting that OM301directly interacts with Bcl-2. Thus, we evaluated the effects of OM301 on mitochondrial function and physiology. Treatment with OM301 decreased mitochondrial membrane potential in different MM cell lines. OM301 also increased mitochondrial superoxide production and induced mitophagy and mitochondrial fission as seen by electron microscopy.
CONCLUSION: Here, we report for the first time that OM301, although designed for p53-selective cells, may instead interact with Bcl-2, which in turn induces mitochondrial dysfunction, leading to cell death irrespective of their TP53 status. Our data suggest that OM301 may be a novel and effective therapeutic option for MM.
Krishnan: REGENERON: Consultancy; MAGENTA: Consultancy; BMS: Consultancy, Current equity holder in publicly-traded company, Speakers Bureau; JANSSEN: Consultancy, Research Funding; City of Hope Cancer Center: Current Employment; SANOFI: Consultancy; GSK: Consultancy; Amgen: Speakers Bureau. Marcucci: Novartis: Other: Speaker and advisory scientific board meetings; Agios: Other: Speaker and advisory scientific board meetings; Abbvie: Other: Speaker and advisory scientific board meetings.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal